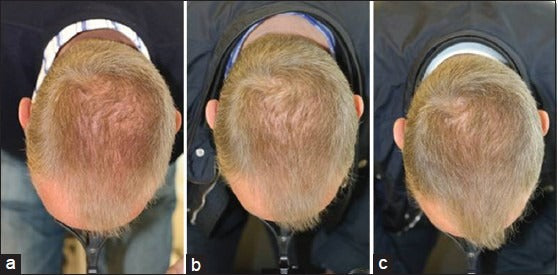
The clinical studies included males & females between the ages of 25-60 yrs. with a diagnosis of androgenetic alopecia who had been experiencing active hair loss within the last 12 months. The inclusion criteria for males required a Norwood- Hamilton classification of IIa to V and Fitzpatrick skin types I to IV. The inclusion criteria for females required a Ludwig (Savin) classification of I-4, II-1, II-2, or frontal.
All subjects were randomized for laser hair loss treatment analysis. A biostatistician calculated the study to be of a proper size to gauge statistically significant results of hair growth and hair density. Subjects were instructed to use the laser hair growth treatment device three times per week on non-consecutive days, 10-15 minutes per treatment for a total of 26 weeks. Hair density measurements were performed at baseline, at 16 and 26 weeks. Additional clinical visits were scheduled to monitor the laser hair loss treatment progress and overall hair growth.
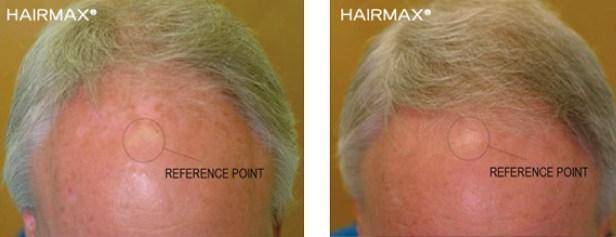
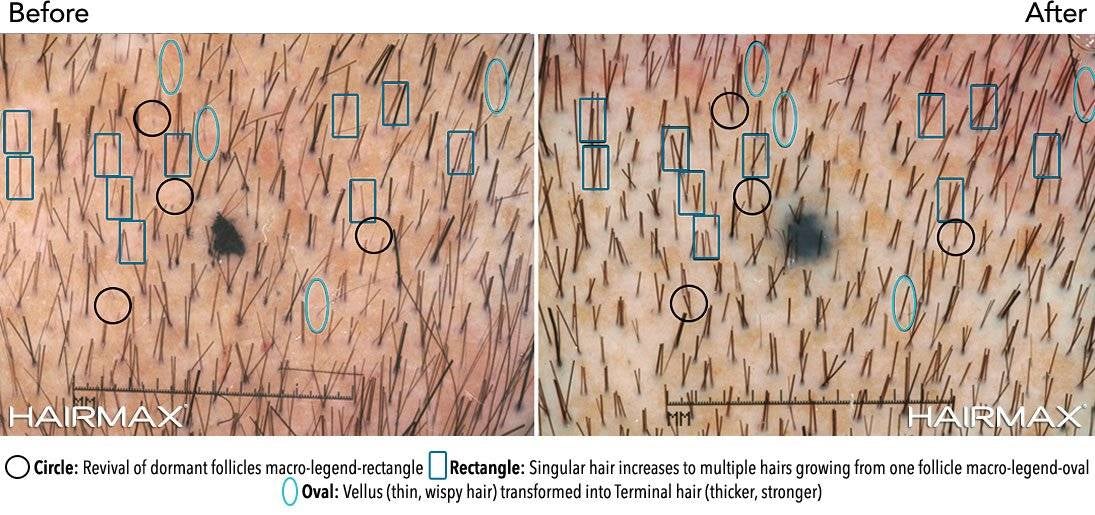
Below are the benefits participants experienced in clinical studies using Hairmax® Laser Devices after 26 weeks of use. (Ongoing treatment is required to maintain these benefits.)